Ongoing Research
Genetics and pathophysiology of MCOPS12
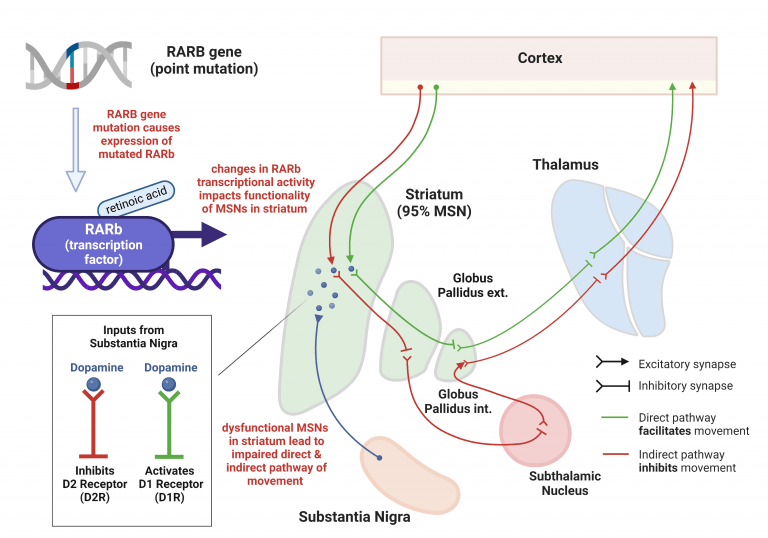
Mutations in the RARB gene, which codes for the retinoic acid receptor beta (RARb) protein, cause a severe and progressive form of early-onset movement disorders (especially dystonia).
RARb is a transcription factor that is activated upon binding to retinoic acid and regulates the expression of a plethora of genes in the human body. One of those targets are a certain type of neurons (i.e. medium spiny neurons) in the striatum, which forms a critical part of the motor control system in the brain. Consequently, movement disorders are typically explained by a dysfunction in the striatum.
Main information input to the striatum comes from glutamatergic neurons from the cortex and dopaminergic neurons from the substantia nigra, which connect to medium spiny neurons (MSNs) in the striatum. The striatum contains two distinct types of MSNs (D1R and D2R) that carry information to different brain regions. Both D1R and D2R contain dopamine receptors and participate in a pivotal neural pathway within the central nervous system to execute and control movement. D1R form part of the direct pathway which facilitates voluntary movement, and D2R are involved in the indirect pathway whose activation leads to the inhibition of movement.
As mentioned, RARb is a transcription factor and the D2R MSNs are one of its targets. Mutations in RARb result in conformational changes of the receptor´s ligand (i.e. retinoic acid) binding domain, hence leading to altered ligand binding and transcriptional activity of the receptor. These could be loss of function, reduction in function, or gain of normal function. It is hypothesized that changes in RARb transcriptional activity impacts MSN gene expression, protein composition, morphology and electrophysiology. Consequently, the direct and indirect pathway of movement becomes impaired thus leading to the observed movement disorders in MCOPS12 patients.
Collaboration with academic research teams
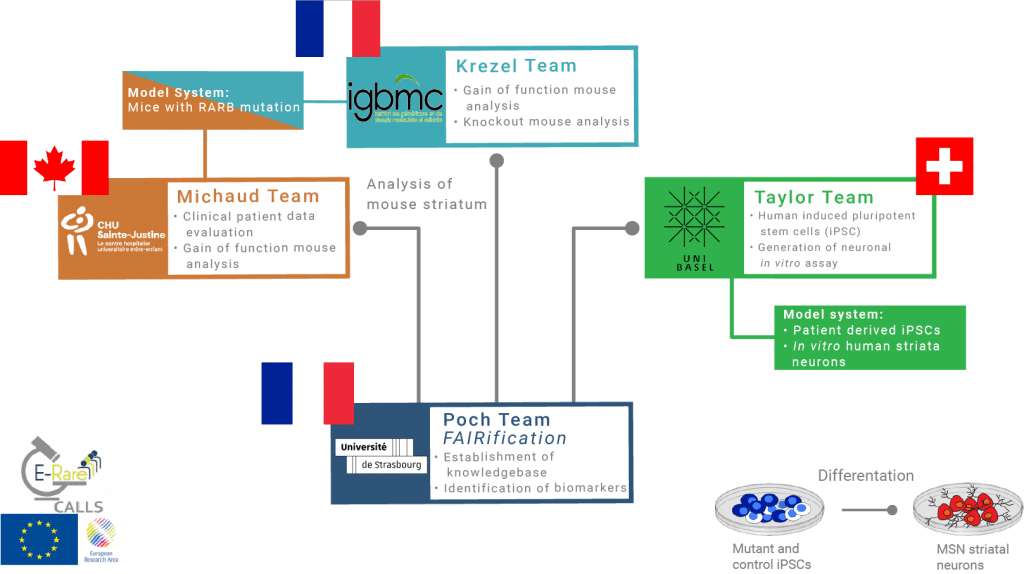
Cure MCOPS12 is collaborating with research teams from four academic institutions (see Figure 3) to establish MCOPS12 disease models, describe and classify disease symptoms (patient natural history study), and determine the pathophysiological mechanism of the movement disorders. Ultimately, the goal is to develop therapeutic approaches for MCOPS12 and robust biomarkers for monitoring the efficiency of these approaches. The teams obtained funding by the European Union and by Cure MCOPS12 to support the research programs.
Core activities
-> Behavioral and histological analyses of RARB transgenic mouse models (having a point mutation equivalent to c.1159C>T (p.R387C) in human patients)
-> Functional genomic, metabolomic and proteomic analyses of transgenic mouse striatum
-> Generation of patient iPSC derived striatal neurons and their transcriptomic, proteomic and morphological analysis
-> Establishment of an integrated knowledgebase with Findable, Accessible, Interoperable and Re-usable (FAIR) research data and identification of biomarkers